Is sodium more reactive than calcium
Is Sodium More Reactive Than Calcium. Calcium cannot displace sodium from its compound because it is less reactive. The reactivity series in order of reactivity. Very reactive calcium metal. Sodium is more reactive.
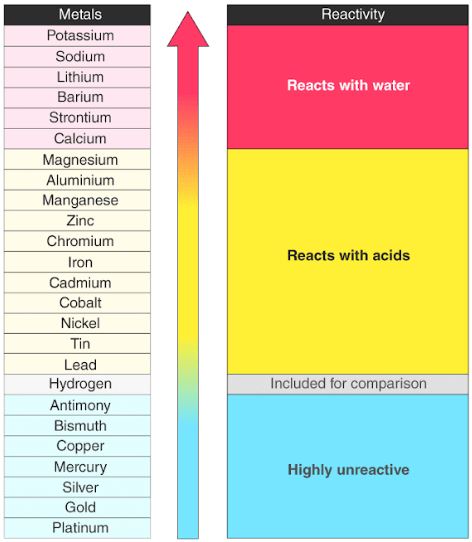 Reactivity Series Reactivity Of Metals Chart Features Uses From byjus.com
Reactivity Series Reactivity Of Metals Chart Features Uses From byjus.com
Why cao is not expected to react with sodium hydroxide. We put then in order of their reactivity in a series that starts with the most reactive. Very reactive calcium metal. Very reactive magnesium metal. Sodium is more reactive. Very reactive sodium metal.
Actually sodium is much more reactive than calcium.
We put then in order of their reactivity in a series that starts with the most reactive. Sodium reacts violently with water to produce aqueous naoh and hydrogen gas. Very reactive calcium metal. This is the reactivity series. Why cao is not expected to react with sodium hydroxide. Some metals are more reactive than others.

Calcium cannot displace sodium from its compound because it is less reactive. Very reactive calcium metal. Substance symbol comment potassium metal. Some metals are more reactive than others. Actually sodium is much more reactive than calcium.
 Source: byjus.com
Source: byjus.com
The reactivity series in order of reactivity. Sodium is more reactive than calcium. Sodium reacts violently with water to produce aqueous naoh and hydrogen gas. Cao is not expected to react with sodium hydroxide because sodium is more reactive than calcium. Why cao is not expected to react with sodium hydroxide.
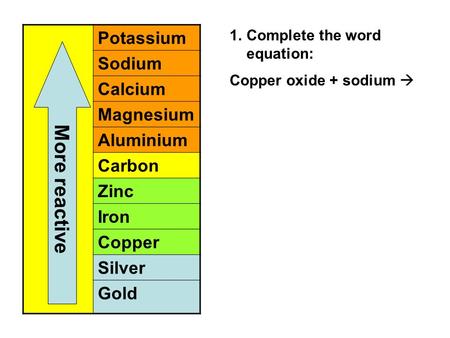 Source: slideplayer.com
Source: slideplayer.com
Why cao is not expected to react with sodium hydroxide. Very reactive sodium metal. Cao is not expected to react with sodium hydroxide because sodium is more reactive than calcium. Some metals are more reactive than others. Substance symbol comment potassium metal.
 Source: quora.com
Source: quora.com
Substance symbol comment potassium metal. Sodium is more reactive than calcium. Very reactive magnesium metal. Calcium cannot displace sodium from its compound because it is less reactive. Very reactive calcium metal.
 Source: quora.com
Source: quora.com
Sodium is more reactive than calcium. Very reactive sodium metal. This is the reactivity series. Sodium is more reactive. Sodium displaces calcium from an ionic compound and hence it is more reactive.
 Source: slideplayer.com
Source: slideplayer.com
The reactivity series in order of reactivity. Sodium displaces calcium from an ionic compound and hence it is more reactive. Actually sodium is much more reactive than calcium. We put then in order of their reactivity in a series that starts with the most reactive. Why cao is not expected to react with sodium hydroxide.
 Source: slideshare.net
Source: slideshare.net
No calcium metal can t reacts with sodium hydroxide because sodium is more reactive than calcium which means sodium has higher tendency to lose electron than calcium that s why calcium can t displace sodium from its aqueous solution. Some metals are more reactive than others. Sodium displaces calcium from an ionic compound and hence it is more reactive. Calcium only reacts with water slowly to produce ca oh 2. We put then in order of their reactivity in a series that starts with the most reactive.
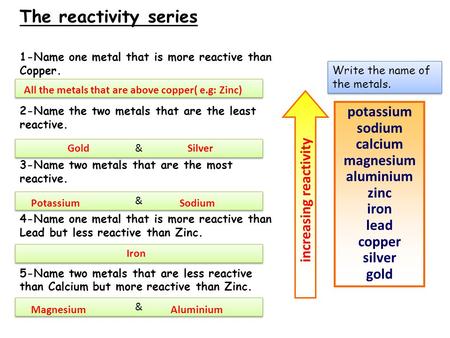 Source: slideplayer.com
Source: slideplayer.com
Sodium is more reactive. Some metals are more reactive than others. No calcium metal can t reacts with sodium hydroxide because sodium is more reactive than calcium which means sodium has higher tendency to lose electron than calcium that s why calcium can t displace sodium from its aqueous solution. Actually sodium is much more reactive than calcium. Sodium reacts violently with water to produce aqueous naoh and hydrogen gas.
 Source: slideshare.net
Source: slideshare.net
Actually sodium is much more reactive than calcium. Very reactive calcium metal. Substance symbol comment potassium metal. Why cao is not expected to react with sodium hydroxide. We put then in order of their reactivity in a series that starts with the most reactive.
 Source: ichemey.blogspot.com
Source: ichemey.blogspot.com
Sodium displaces calcium from an ionic compound and hence it is more reactive. Sodium reacts violently with water to produce aqueous naoh and hydrogen gas. Very reactive sodium metal. Actually sodium is much more reactive than calcium. Very reactive magnesium metal.
 Source: quora.com
Source: quora.com
The reactivity series in order of reactivity. Some metals are more reactive than others. Very reactive magnesium metal. Why cao is not expected to react with sodium hydroxide. Calcium cannot displace sodium from its compound because it is less reactive.
 Source: quora.com
Source: quora.com
Very reactive sodium metal. Calcium cannot displace sodium from its compound because it is less reactive. Actually sodium is much more reactive than calcium. Very reactive magnesium metal. The reactivity series in order of reactivity.
 Source: quora.com
Source: quora.com
No calcium metal can t reacts with sodium hydroxide because sodium is more reactive than calcium which means sodium has higher tendency to lose electron than calcium that s why calcium can t displace sodium from its aqueous solution. We put then in order of their reactivity in a series that starts with the most reactive. Sodium is more reactive. Cao is not expected to react with sodium hydroxide because sodium is more reactive than calcium. The reactivity series in order of reactivity.
 Source: richardbowles.co.uk
Source: richardbowles.co.uk
Actually sodium is much more reactive than calcium. Very reactive sodium metal. Actually sodium is much more reactive than calcium. Calcium only reacts with water slowly to produce ca oh 2. Very reactive calcium metal.
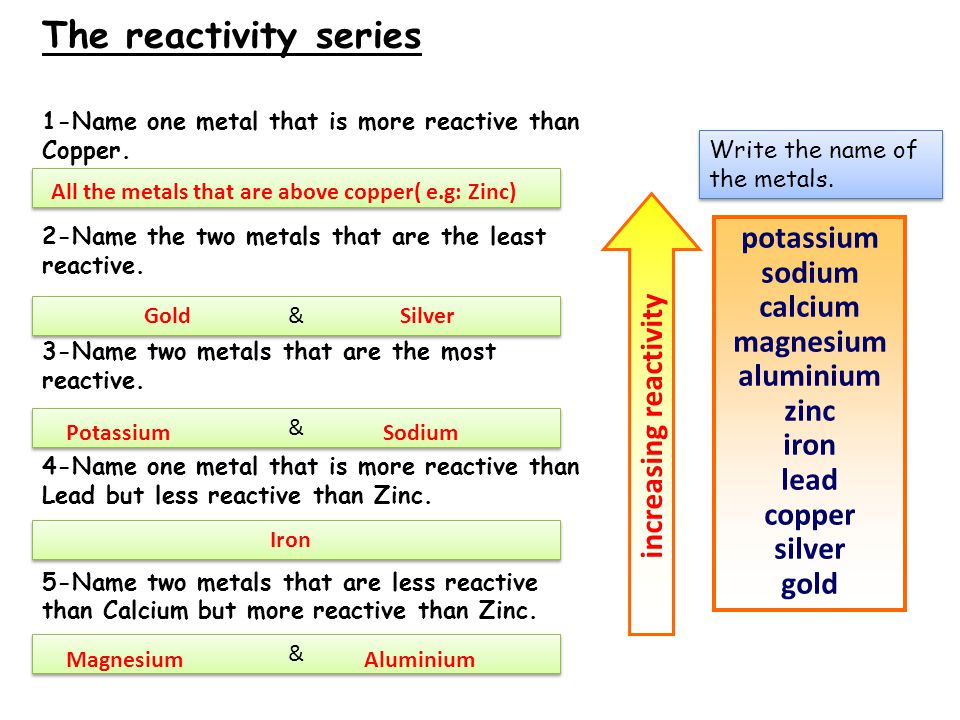 Source: slideplayer.com
Source: slideplayer.com
Very reactive sodium metal. Sodium displaces calcium from an ionic compound and hence it is more reactive. Very reactive sodium metal. This is the reactivity series. Very reactive calcium metal.
If you find this site value, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title is sodium more reactive than calcium by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.