Phenolphthalein ph indicator
Phenolphthalein Ph Indicator. In acidic solutions it is colorless. The quantity of indicator in aqueous aq or alcohol alc solution is specified. Litmus phenolphthalein and methyl orange are all indicators that are commonly used in the. Besides from 0 to 6 9 resides all the acids and the less the number of acid the more acidic they are.
 Phenolphthalein 77 09 8 From chemicalbook.com
Phenolphthalein 77 09 8 From chemicalbook.com
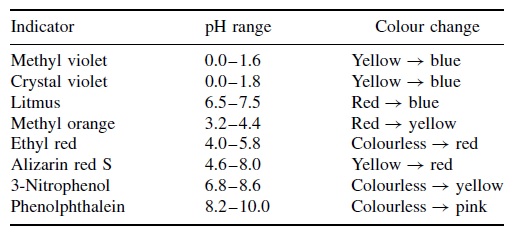
In acidic solutions it is colorless. Phenolphthalein adopts at least four different states in aqueous solution as a result of ph changes. It also serves as a component of universal indicator together with methyl red bromothymol blue and thymol blue. Several acid base indicators are listed below some more than once if they can be used over multiple ph ranges. Indicators are substances that change colour when they are added to acidic or alkaline solutions. Packaging 100 500 g in poly bottle.
Phenolphthalein c 20 h 14 o 4 an organic compound of the phthalein family that is widely employed as an acid base indicator.
Phenolphthalein is another commonly used indicator for titrations and is another weak acid. Besides from 0 to 6 9 resides all the acids and the less the number of acid the more acidic they are. Phenolphthalein is also used as a model organic compound in the evaluation of the capture efficiency of various cyclodextrin constructs such as nanofiber cd. Common acid base indicators. Packaging 100 500 g in poly bottle. Phenolphthalein is used as a general ph indicator.
 Source: amazon.in
Source: amazon.in
Indicators are substances that change colour when they are added to acidic or alkaline solutions. Phenolphthalein exerts laxative effects by stimulating the intestinal mucosa and constricting smooth muscles. Phenolphthalein is another commonly used indicator for titrations and is another weak acid. The phenolphthalein indicator allows chemists to visually identify whether a substance is an acid or a base. The quantity of indicator in aqueous aq or alcohol alc solution is specified.
 Source: chemistry.elmhurst.edu
Source: chemistry.elmhurst.edu
How does phenolphthalein indicator work. The phenolphthalein indicator allows chemists to visually identify whether a substance is an acid or a base. As an indicator of a solution s ph phenolphthalein is colourless below ph 8 5 and attains a pink to deep red hue above ph 9 0. Phenolphthalein exerts laxative effects by stimulating the intestinal mucosa and constricting smooth muscles. Adding extra hydrogen ions shifts the position of equilibrium to the left and turns the indicator colourless.
 Source: quora.com
Source: quora.com
Phenolphthalein adopts at least four different states in aqueous solution as a result of ph changes. Phenolphthalein is also used as a model organic compound in the evaluation of the capture efficiency of various cyclodextrin constructs such as nanofiber cd. It is colorless in acidic solutions and turns pink in basic solutions. Phenolphthalein is an organic compound used as a laboratory reagent and ph indicator. However phenolphthalein is no longer used as a laxative due to the suspected carcinogenicity of this compound.
 Source: sciencecompany.com
Source: sciencecompany.com
In acidic solutions it is colorless. While the ph 7 is neutral which is the ph of water. Phenolphthalein c 20 h 14 o 4 an organic compound of the phthalein family that is widely employed as an acid base indicator. Litmus phenolphthalein and methyl orange are all indicators that are commonly used in the. The quantity of indicator in aqueous aq or alcohol alc solution is specified.
 Source: indiamart.com
Source: indiamart.com
Phenolphthalein c 20 h 14 o 4 an organic compound of the phthalein family that is widely employed as an acid base indicator. In this case the weak acid is colourless and its ion is bright pink. Adding extra hydrogen ions shifts the position of equilibrium to the left and turns the indicator colourless. It also serves as a component of universal indicator together with methyl red bromothymol blue and thymol blue. The phenolphthalein indicator allows chemists to visually identify whether a substance is an acid or a base.

Phenolphthalein is also used as a model organic compound in the evaluation of the capture efficiency of various cyclodextrin constructs such as nanofiber cd. In acidic solutions it is colorless. The ph scale has a marking from 0 to 14 fro detecting acid and base. Litmus phenolphthalein and methyl orange are all indicators that are commonly used in the. Phenolphthalein exerts laxative effects by stimulating the intestinal mucosa and constricting smooth muscles.
 Source: sciencecompany.com
Source: sciencecompany.com
Phenolphthalein is an organic compound used as a laboratory reagent and ph indicator. Several acid base indicators are listed below some more than once if they can be used over multiple ph ranges. In acidic solutions it is colorless. It is colorless in acidic solutions and turns pink in basic solutions. It also serves as a component of universal indicator together with methyl red bromothymol blue and thymol blue.
 Source: flexbooks.ck12.org
Source: flexbooks.ck12.org
Common acid base indicators. Adding extra hydrogen ions shifts the position of equilibrium to the left and turns the indicator colourless. The quantity of indicator in aqueous aq or alcohol alc solution is specified. It is colorless in acidic solutions and turns pink in basic solutions. In this case the weak acid is colourless and its ion is bright pink.
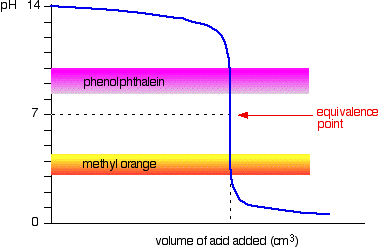 Source: chemguide.co.uk
Source: chemguide.co.uk
Indicators are substances that change colour when they are added to acidic or alkaline solutions. In alkaline it turns pink. It also serves as a component of universal indicator together with methyl red bromothymol blue and thymol blue. Adding extra hydrogen ions shifts the position of equilibrium to the left and turns the indicator colourless. Phenolphthalein is also used as a model organic compound in the evaluation of the capture efficiency of various cyclodextrin constructs such as nanofiber cd.
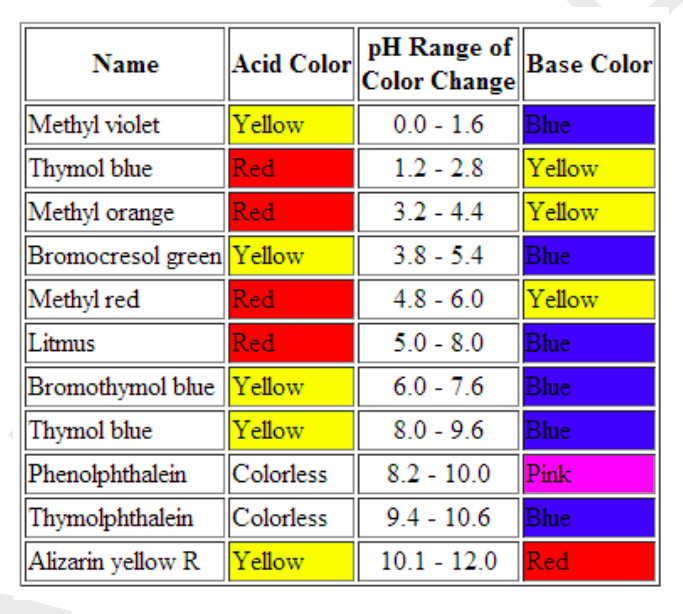 Source: chegg.com
Source: chegg.com
Indicators are substances that change colour when they are added to acidic or alkaline solutions. Phenolphthalein adopts at least four different states in aqueous solution as a result of ph changes. Phenolphthalein exerts laxative effects by stimulating the intestinal mucosa and constricting smooth muscles. Phenolphthalein is used as a general ph indicator. Phenolphthalein is another commonly used indicator for titrations and is another weak acid.
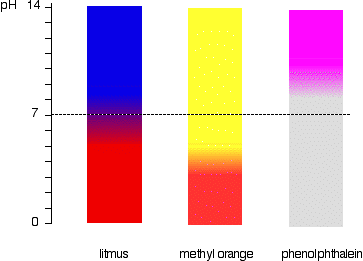 Source: chemguide.co.uk
Source: chemguide.co.uk
Phenolphthalein is an organic compound used as a laboratory reagent and ph indicator. Phenolphthalein s common use is as an indicator in acid base titrations. The color change in phenolphthalein is a result of ionization and this alters the shape of the phenolphthalein molecules. Phenolphthalein c 20 h 14 o 4 an organic compound of the phthalein family that is widely employed as an acid base indicator. The ph scale has a marking from 0 to 14 fro detecting acid and base.
 Source: quora.com
Source: quora.com
Packaging 100 500 g in poly bottle. How does phenolphthalein indicator work. Phenolphthalein is used as a general ph indicator. Common acid base indicators. In acidic solutions it is colorless.
 Source: compoundchem.com
Source: compoundchem.com
The phenolphthalein indicator allows chemists to visually identify whether a substance is an acid or a base. Litmus phenolphthalein and methyl orange are all indicators that are commonly used in the. In alkaline it turns pink. While the ph 7 is neutral which is the ph of water. Phenolphthalein is another commonly used indicator for titrations and is another weak acid.
 Source: chemicalbook.com
Source: chemicalbook.com
Litmus phenolphthalein and methyl orange are all indicators that are commonly used in the. Indicators are substances that change colour when they are added to acidic or alkaline solutions. The color change in phenolphthalein is a result of ionization and this alters the shape of the phenolphthalein molecules. Phenolphthalein c 20 h 14 o 4 an organic compound of the phthalein family that is widely employed as an acid base indicator. Besides phenolphthalein structure form of the carbon hydrogen and oxygen chains.
 Source: amazon.in
Source: amazon.in
The color change in phenolphthalein is a result of ionization and this alters the shape of the phenolphthalein molecules. Tried and true indicators include thymol blue tropeolin oo methyl yellow methyl orange bromphenol blue bromcresol green methyl red bromthymol blue phenol red. Indicators are substances that change colour when they are added to acidic or alkaline solutions. Phenolphthalein adopts at least four different states in aqueous solution as a result of ph changes. While the ph 7 is neutral which is the ph of water.
If you find this site adventageous, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title phenolphthalein ph indicator by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.