What is the meaning of distillation in science
What Is The Meaning Of Distillation In Science. It s based on the different boiling point or volatility values of the components. Distillation is defined as a refining process that collects the pure liquid or vapors given off by heating and then cooling a mixture. Distillation can be used to separate two liquids that are fully mixed together to form a solution. Distillation is the process that occurs when a liquid sample is volatilized to produce a vapor that is subsequently condensed to a liquid richer in the more volatile components of the original sample.
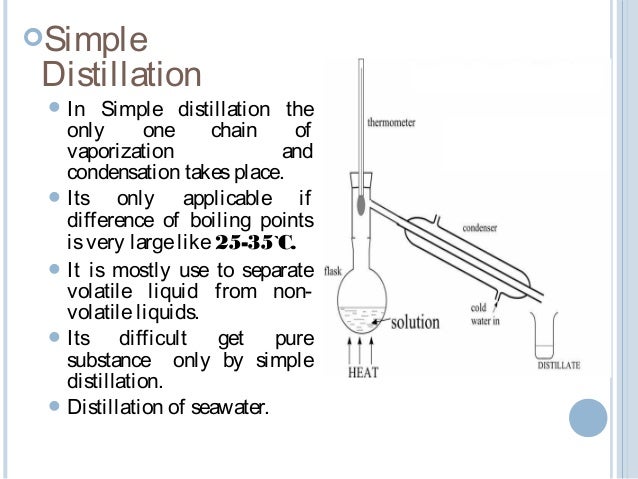 Distillation From slideshare.net
Distillation From slideshare.net
We cover the cycle of vaporization cooling and condensation that is the basis for the. Distillation is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation. An example of distillation is how vodka is made from a grain. The heart is the part of a distillate produced during distillation that is separated and kept to make alcoholic beverages. When two liquids can fully mix together they are called miscible liquids. Distillation may result in essentially complete separation nearly pure components or it may be a partial separation that increases the concentration of selected components in the mixture.
Distillation definition the volatilization or evaporation and subsequent condensation of a liquid as when water is boiled in a retort and the steam is condensed in a cool receiver.
When two liquids can fully mix together they are called miscible liquids. Distillation may result in essentially complete separation nearly pure components or it may be a partial separation that increases the concentration of selected components in the mixture. Distillation is the process that occurs when a liquid sample is volatilized to produce a vapor that is subsequently condensed to a liquid richer in the more volatile components of the original sample. Distillation definition distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture. It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. Distillation the process involving the conversion of a liquid into vapor that is subsequently condensed back to liquid form.
 Source: eschool.iaspaper.net
Source: eschool.iaspaper.net
Distillation can be used to separate two liquids that are fully mixed together to form a solution. Distillation definition the volatilization or evaporation and subsequent condensation of a liquid as when water is boiled in a retort and the steam is condensed in a cool receiver. Science primary school what is the meaning of distillation 2 see answers amankumar1712003 amankumar1712003 explanation. It s based on the different boiling point or volatility values of the components. Distillation is the process that occurs when a liquid sample is volatilized to produce a vapor that is subsequently condensed to a liquid richer in the more volatile components of the original sample.
 Source: byjus.com
Source: byjus.com
It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. Science primary school what is the meaning of distillation 2 see answers amankumar1712003 amankumar1712003 explanation. The technique may be used to separate components of a mixture or to aid in purification. The volatilization process usually involves heating the liquid but it may also be achieved by reducing the pressure or by a combination of both.
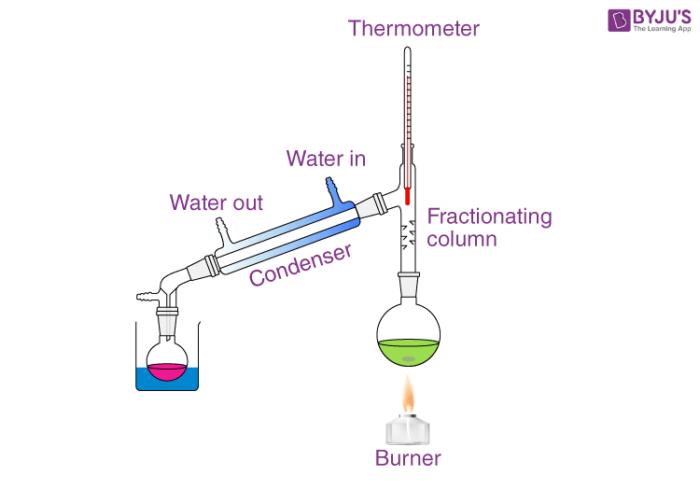 Source: byjus.com
Source: byjus.com
It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. Liquids that do not mix. Science primary school what is the meaning of distillation 2 see answers amankumar1712003 amankumar1712003 explanation. An example of distillation is how vodka is made from a grain.
 Source: docbrown.info
Source: docbrown.info
Distillation is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation. When two liquids can fully mix together they are called miscible liquids. Distillation can be used to separate two liquids that are fully mixed together to form a solution. Distillation the process involving the conversion of a liquid into vapor that is subsequently condensed back to liquid form. We cover the cycle of vaporization cooling and condensation that is the basis for the.
 Source: en.wikipedia.org
Source: en.wikipedia.org
Distillation definition the volatilization or evaporation and subsequent condensation of a liquid as when water is boiled in a retort and the steam is condensed in a cool receiver. Liquids that do not mix. Distillation may result in essentially complete separation nearly pure components or it may be a partial separation that increases the concentration of selected components in the mixture. Distillation the process involving the conversion of a liquid into vapor that is subsequently condensed back to liquid form. When two liquids can fully mix together they are called miscible liquids.
 Source: youtube.com
Source: youtube.com
The technique may be used to separate components of a mixture or to aid in purification. Distillation is defined as a refining process that collects the pure liquid or vapors given off by heating and then cooling a mixture. Liquids that do not mix. To separate a mixture of liquids the liquid can be heated to force components which have different boiling points into the gas phase. When two liquids can fully mix together they are called miscible liquids.
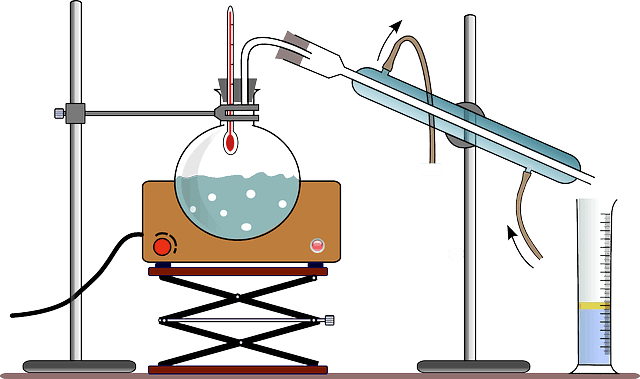 Source: eartheclipse.com
Source: eartheclipse.com
Distillation the process involving the conversion of a liquid into vapor that is subsequently condensed back to liquid form. It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. The substances which make up the other parts of the distillation have an unpleasant odour or taste and often are harmful to human health. We cover the cycle of vaporization cooling and condensation that is the basis for the. Liquids that do not mix.
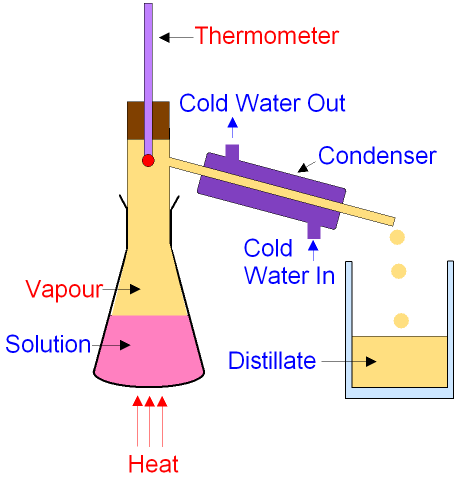 Source: gcsescience.com
Source: gcsescience.com
Distillation definition distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture. Distillation may result in essentially complete separation nearly pure components or it may be a partial separation that increases the concentration of selected components in the mixture. It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points. Distillation is the technique of heating a liquid to create vapor which is collected when cooled separate from the original liquid. Distillation can be used to separate two liquids that are fully mixed together to form a solution.
 Source: en.wikipedia.org
Source: en.wikipedia.org
Distillation is the technique of heating a liquid to create vapor which is collected when cooled separate from the original liquid. Distillation may result in essentially complete separation nearly pure components or it may be a partial separation that increases the concentration of selected components in the mixture. It s based on the different boiling point or volatility values of the components. Distillation definition distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture. Distillation is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation.
 Source: thoughtco.com
Source: thoughtco.com
Distillation is the technique of heating a liquid to create vapor which is collected when cooled separate from the original liquid. Put simply it is the good tasting part of the distillation that is safe to digest. Distillation is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation. When two liquids can fully mix together they are called miscible liquids. Distillation definition the volatilization or evaporation and subsequent condensation of a liquid as when water is boiled in a retort and the steam is condensed in a cool receiver.
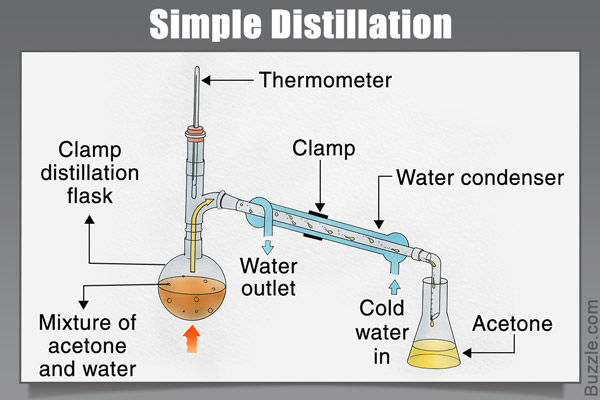 Source: sciencestruck.com
Source: sciencestruck.com
An example of distillation is how vodka is made from a grain. An example of distillation is how vodka is made from a grain. Science primary school what is the meaning of distillation 2 see answers amankumar1712003 amankumar1712003 explanation. When two liquids can fully mix together they are called miscible liquids. The substances which make up the other parts of the distillation have an unpleasant odour or taste and often are harmful to human health.
 Source: slideshare.net
Source: slideshare.net
Put simply it is the good tasting part of the distillation that is safe to digest. Distillation the process involving the conversion of a liquid into vapor that is subsequently condensed back to liquid form. An example of distillation is how vodka is made from a grain. Put simply it is the good tasting part of the distillation that is safe to digest. It s based on the different boiling point or volatility values of the components.
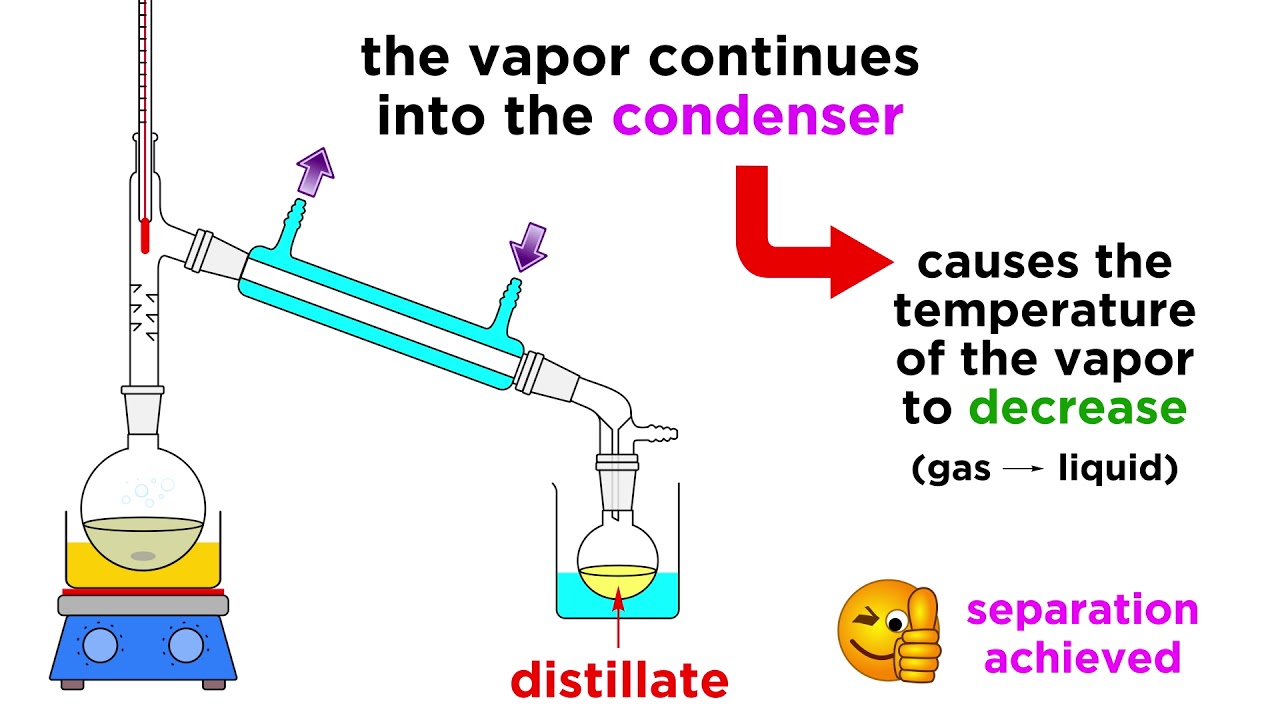 Source: m.youtube.com
Source: m.youtube.com
The heart is the part of a distillate produced during distillation that is separated and kept to make alcoholic beverages. Explore distillation the chemist s most useful method for separating substances dissolved in liquids. Distillation can be used to separate two liquids that are fully mixed together to form a solution. The technique may be used to separate components of a mixture or to aid in purification. An example of distillation is how vodka is made from a grain.

Distillation definition distillation is a widely used method for separating mixtures based on differences in the conditions required to change the phase of components of the mixture. The volatilization process usually involves heating the liquid but it may also be achieved by reducing the pressure or by a combination of both. Distillation can be used to separate two liquids that are fully mixed together to form a solution. Distillation is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation. Distillation is the process that occurs when a liquid sample is volatilized to produce a vapor that is subsequently condensed to a liquid richer in the more volatile components of the original sample.
 Source: simple.wikipedia.org
Source: simple.wikipedia.org
Distillation definition the volatilization or evaporation and subsequent condensation of a liquid as when water is boiled in a retort and the steam is condensed in a cool receiver. Liquids that do not mix. Distillation is the process that occurs when a liquid sample is volatilized to produce a vapor that is subsequently condensed to a liquid richer in the more volatile components of the original sample. Distillation definition the volatilization or evaporation and subsequent condensation of a liquid as when water is boiled in a retort and the steam is condensed in a cool receiver. It is used to separate liquids from nonvolatile solids or in the separation of two or more liquids having different boiling points.
If you find this site beneficial, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title what is the meaning of distillation in science by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.